Cro services in
Liver (MASH)
Gubra is industry-leading in translational models of metabolic dysfunction-associated steatohepatitis (MASH) and MASH-driven hepatocellular carcinoma (MASH-HCC). Our automated AI-based histology pipeline enables effective, accurate and objective analysis of drug effects on pivotal clinical endpoints.
Why Gubra?
- Expert scientific guidance in MASH
- Clinically translatable models
- GAN DIO-MASH mouse ranked #1 by LITMUS consortium
- Biopsy-enabled stratification of baseline disease
- Profile compounds using clinical trial endpoints
- Tailored study design and rapid initiation


Consult with Michael Feigh
Vice President, Scientific Sales
Choose the right animal model for your MASH-targeted drug candidate.
What is MASH?
Metabolic Dysfunction-associated Steatohepatitis (MASH)
Metabolic dysfunction-associated steatohepatitis (MASH) and metabolic dysfunction-associated steatotic liver disease (MASLD), respectively, are now the replacement terms for non-alcoholic steatohepatitis (NASH) and non-alcoholic fatty liver disease (NAFLD).
Industry-leading models for translatability into the clinic
Choosing the right translational model of MASH is crucial for efficacy evaluation and bridging to clinical development. Our GAN DIO-MASH mouse model has been ranked #1 by the LITMUS consortium for its human proximity score and clinical translatability. Our model is already induced and ready to be used for your study. With reference to standard clinical practice, all animals are biopsy-confirmed for stratification of baseline disease which allows for within-subject evaluation of histological outcomes.
Main Offerings
Full metabolic
assessment
Biochemical assessment
liver & plasma
NAS & fibrosis scoring
(pre-post)
Quantitative histology
Steatohepatitis & fibrosis
Hepatic transcriptomic
profile
GAN DIO-MASH
mice readily available
High clinical translatability
Models of MASH & MASH-HCC
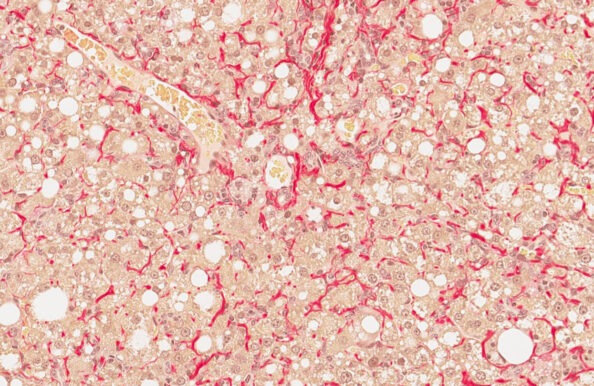
GAN DIO-MASH mouse
- Ranked #1 for human translatability by the LITMUS consortium
- Diet-induced obesity with metabolic disease
- Fibrosing MASH ≥28 weeks of GAN dieting
- Biopsy-confirmed MASH and fibrosis
- Clinical histopathological scoring (pre-post)
- Validated with clinical late-stage drugs
- Therapeutic intervention
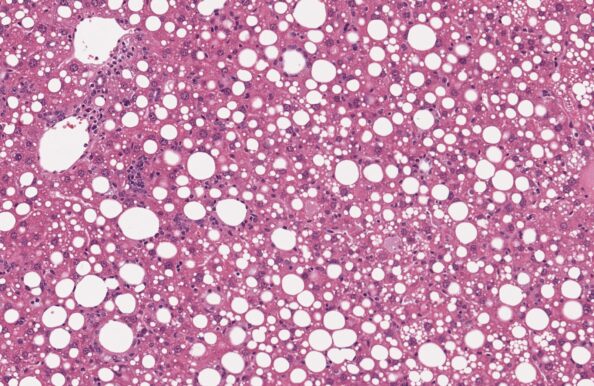
GAN DIO-MASH-HCC mouse
- Extended GAN DIO-MASH model
- HCC development ≥ 48 weeks of GAN diet feeding
- Biopsy-confirmed MASH and fibrosis
- Clinical histopathological scoring (pre-post)
- Macroscopic HCC evaluation
- Validated with clinical late-stage drugs
- Therapeutic/Prophylactic intervention
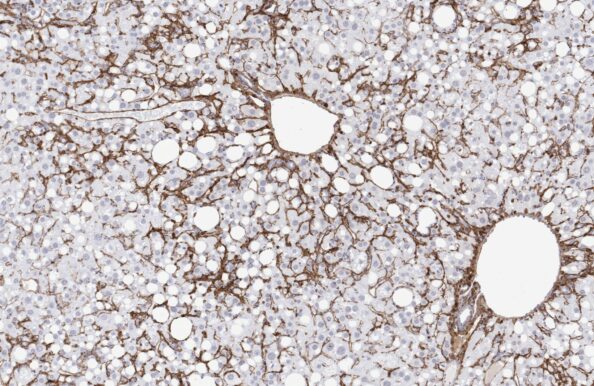
GAN ob/ob-MASH mouse
- Top-ranked for human translatability by the LITMUS consortium
- Accelerated MASH with progressive advanced fibrosis
- GAN diet feeding for ≥12 weeks
- Genetic obesity with severe metabolic disease
- Clinical histopathological scoring
- Validated with clinical late-stage drugs
- Therapeutic/Prophylactic intervention
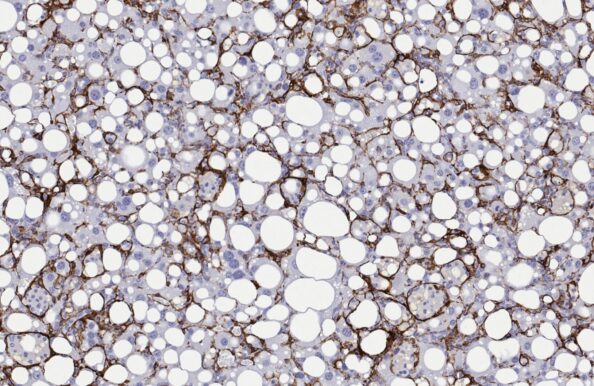
CDAA-HFD mouse
- Recommended by the LITMUS consortium
- Accelerated MASH with progressive advanced fibrosis
- CDAA-HFD administration for ≥12 weeks
- Non-obese phenotype
- Clinical histopathological scoring (post)
- Macroscopic tumor evaluation
- Validated with clinical late-stage drugs
- Therapeutic/Prophylactic intervention
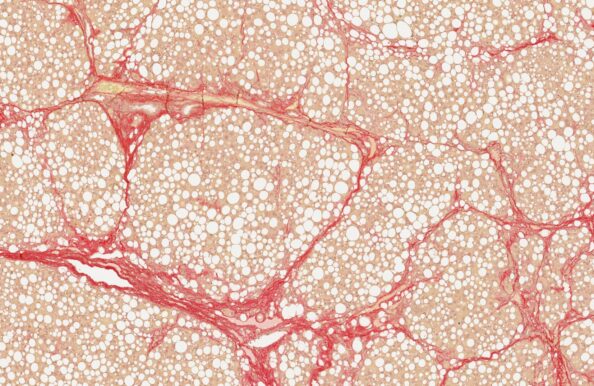
CDAA-HFD rat
- Accelerated MASH with progression to cirrhosis
- CDAA-HFD administration for up to 16 weeks
- Non-obese phenotype
- Biopsy-confirmed MASH and fibrosis
- Clinical histopathological scoring (pre-post)
- Macroscopic tumor evaluation
- Validated with clinical late-stage drugs
- Therapeutic/Prophylactic intervention
Related pages
For further information
Contact us
Gubra
Hørsholm Kongevej 11B
2970 Hørsholm
Denmark
+45 3152 2650